Teaching students to understand chemistry and instructing a student to be a trained chemist require different strategies of teaching.
The chemist wants to achieve an end-product or make valid measurements as efficiently as possible. For that, in the laboratory, we have heaters, stirrers, and quite large and sophisticated pieces of equipment from burettes to Gas Liquid Chromatography linked to mass spectrometry. A chemistry teacher though has a different aim.
The teachers' aim is ensure students understand chemistry with the ultimate aim to (yes, pass the exam), inspire students to become chemists, biochemists, pharmacists, technicians, engineers, medics, etc. If the student wishes to go into non-science careers, they will take with them a knowledge of methodology to give them a good scientific background and show them how essential a knowledge of chemistry is to understand the modern world..
This is why some of the microscale techniques outlined below are not what you find in a student text book when starting chemistry. Many text books repeat the same standard experiments the authors did when teaching or being taught. The experiments are merely a side show, not an integral part of learning.
The chemist wants to achieve an end-product or make valid measurements as efficiently as possible. For that, in the laboratory, we have heaters, stirrers, and quite large and sophisticated pieces of equipment from burettes to Gas Liquid Chromatography linked to mass spectrometry. A chemistry teacher though has a different aim.
The teachers' aim is ensure students understand chemistry with the ultimate aim to (yes, pass the exam), inspire students to become chemists, biochemists, pharmacists, technicians, engineers, medics, etc. If the student wishes to go into non-science careers, they will take with them a knowledge of methodology to give them a good scientific background and show them how essential a knowledge of chemistry is to understand the modern world..
This is why some of the microscale techniques outlined below are not what you find in a student text book when starting chemistry. Many text books repeat the same standard experiments the authors did when teaching or being taught. The experiments are merely a side show, not an integral part of learning.
Alex Johnstone (see previous blog) was onto this in his books “Chemistry Takes Shape” and “Chemistry About Us” (see extract from preface below) in the 60s and 70s. His research backs up his original observations on short term working memory, teaching from evidence and altering the sequence to fit the day to day contact of students with chemistry in their environment; they are hardly applied in teaching schemes of work today.
In Chemistry About Us, organic chemistry is at the forefront because it deals with molecules and not ions. pH is introduced three quarters of the way through, because he observed the students found it difficult. Now I see students at GCSE in the UK do not study logarithms so that makes it even worse to understand the scale.
In Chemistry About Us, organic chemistry is at the forefront because it deals with molecules and not ions. pH is introduced three quarters of the way through, because he observed the students found it difficult. Now I see students at GCSE in the UK do not study logarithms so that makes it even worse to understand the scale.
Starting with the solid
Teachers are far too nice to students; we provide them with chemicals so that all they have to do is mix them and see what happens. There is of course a need to do this. Practical lessons are stressful to the teacher; they are constantly on the lookout for safety issues or students requesting help. A lesson would be greatly slowed down if students had to prepare a solution (and we would demand a concentration wouldn’t we). Solids are often slow to dissolve at room temperature so heating would be required. Practical work is often confusing to the student; it overloads their short-term working memory so that they lose the ultimate aim of what they are doing. This can lead to poor behaviour. Some students only like practical work because it appears to them to be “not proper work”, ie, not writing, not doing sums, not real thinking.
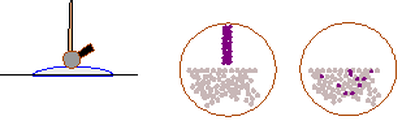
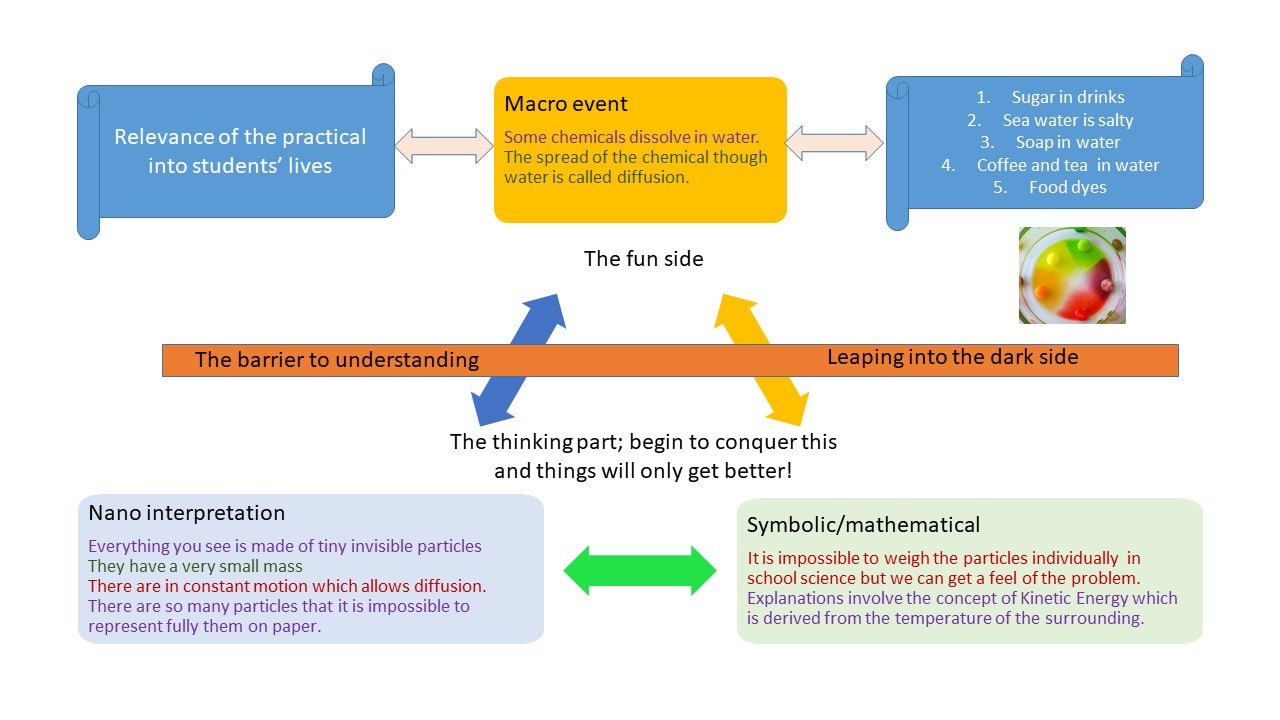
Using the Johnstone triangle, studying chemistry required thinking at the invisible nano-level, thinking in models that are always to be improved, refined, symbolic notations, and achieving quantitative data. This upsets people who just want the right answer.
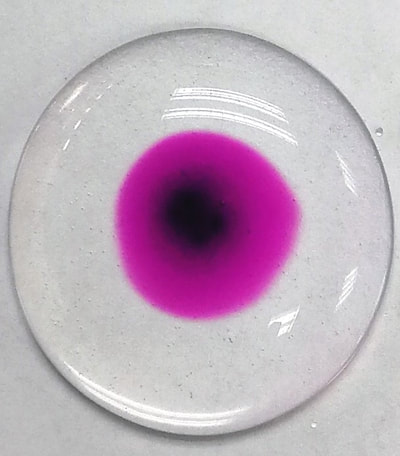
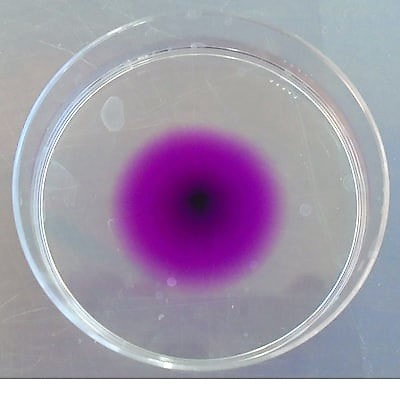
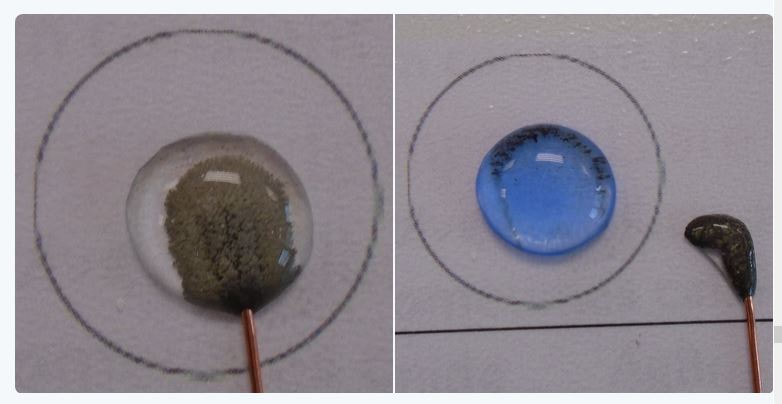
A puddle of water about 1.5cm in diameter is placed on plastic sheet folder. A toothpick is wetted, added to a little solid potassium manganate(VII) and then dabbed into the centre of puddle. It can also be carried out in 10 ml of water in a Petri dish (picture right).
Teachers are far too nice to students; we provide them with chemicals so that all they have to do is mix them and see what happens. There is of course a need to do this. Practical lessons are stressful to the teacher; they are constantly on the lookout for safety issues or students requesting help. A lesson would be greatly slowed down if students had to prepare a solution (and we would demand a concentration wouldn’t we). Solids are often slow to dissolve at room temperature so heating would be required. Practical work is often confusing to the student; it overloads their short-term working memory so that they lose the ultimate aim of what they are doing. This can lead to poor behaviour. Some students only like practical work because it appears to them to be “not proper work”, ie, not writing, not doing sums, not real thinking.
Using the Johnstone triangle, studying chemistry required thinking at the invisible nano-level, thinking in models that are always to be improved, refined, symbolic notations, and achieving quantitative data. This upsets people who just want the right answer.
A puddle of water about 1.5cm in diameter is placed on plastic sheet folder. A toothpick is wetted, added to a little solid potassium manganate(VII) and then dabbed into the centre of puddle. It can also be carried out in 10 ml of water in a Petri dish (picture right).
This is a well-known diffusion experiment which many will think of as trivial, carried out for 11-12 years olds in the UK. The very tiny particles of water and potassium manganate(VII) (ions not introduced yet) are moving around powered by the kinetic energy they receive from heat of the surroundings. By using the solid, the particles of water and potassium manganate are doing the work, not you, because you are not stirring! At this age, the young student brain cannot really take in the full significance of what is happening. These experiments will need to be repeated continuously; but are they? Diffusion is so important in chemistry and biology, yet it is often given a cursory note in text books and never mentioned again.
The problem is that the macro event
You can ask (or do it yourself) start for a storyline cartoon of what is happening.
The problem is that the macro event
- involves tiny particles invisible to the naked eye,
- involves huge numbers of particles, far more than we can imagine,
- relies on many interlinked variables, (temperature, solubility, nature of solvents etc),
- depends on being able to integrate with other concepts, (ions molecules, potential energy etc), and
- involves mathematical concepts.
You can ask (or do it yourself) start for a storyline cartoon of what is happening.
What about a nano-scale interpretation storyline cartoon? It is as though we put a powerful magnifying glass against the events at the end of the toothpick. This is an example of modelling. One has to be realistic about modelling in science terms. It is not the perfect truth and any subsequent refinements of our particle model must take into account previous events.

Can we do any simple arithmetic with this to get a feel of the scale. The procedure can be carried out in 5cm diameter Petri dish left (takes 15 minutes to dissolve and spread out) or stirred (why not, be a chemist!).
The mass of potassium manganate(VII) on the end of the toothpick is in the order of 0.005g of solid and this dissolves in 10 ml of water. In a 3ml-pipette there are 20 drops to the ml and you can add 1 drop of this purple solution to another 10 ml of water and still see the colour from the side when mixed (see photo on the right). There is now 0.000025g of potassium manganate(VII) in 10 ml and I could pick up a drop of that in the pipette and although we cannot see the colour, there must be some potassium manganate(VII) there. (There is actually 0.000000125g but we chemists know that in that drop still a huge number of manganate(VII) particles, about 470,000,000,000,000 in 30100000000000000000000 particle of water, ie 1 purple particle surrounded by 67 million water molecules).
Here are some chemical reactions starting with the solid
The mass of potassium manganate(VII) on the end of the toothpick is in the order of 0.005g of solid and this dissolves in 10 ml of water. In a 3ml-pipette there are 20 drops to the ml and you can add 1 drop of this purple solution to another 10 ml of water and still see the colour from the side when mixed (see photo on the right). There is now 0.000025g of potassium manganate(VII) in 10 ml and I could pick up a drop of that in the pipette and although we cannot see the colour, there must be some potassium manganate(VII) there. (There is actually 0.000000125g but we chemists know that in that drop still a huge number of manganate(VII) particles, about 470,000,000,000,000 in 30100000000000000000000 particle of water, ie 1 purple particle surrounded by 67 million water molecules).
Here are some chemical reactions starting with the solid

The precipitation of silver or lead iodide
I choose this for students to do because the precipitate is coloured and yet the reagents are colourless.
- Place a polypropylene sheet on the on the flat table with a sheet of white paper beneath.
- Place a few grains of two solids of chemicals you know give a precipitate eg, silver nitrate and potassium iodide.
- Using a pipette make a puddle of water about 2cm in diameter, between the grains
- Use a wooden spill from either end move some grains into each side of the puddle.
- Wait as the solids dissolve, the particles move and, when they meet up down he centre, a yellow solid “precipitate” forms.
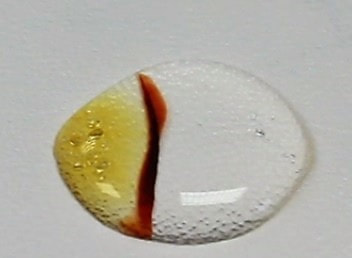
The picture below shows the formation of the iron/thiocyanate complex carried out in the same manner using iron(III) nitrate and potassium thiocyanate.
Displacement
- Place a polypropylene sheet on the on the flat table with a sheet of white paper beneath.
- Place a few small crystals colourless silver nitrate crystals on the sheet.
- Make a 2cm diameter puddle of pure water about 1 cm from the crystals.
- Move the colourless solid into the puddle and let the solid dissolve (the less you use the quicker, it will work!)
- Insert a small piece of copper wire into the water.
- To see that copper has dissolved from the wire, add a drop of 1M ammonia solution and the blue colour suddenly appears.
Neutralization
- Place a polypropylene sheet on the on the flat table with a sheet of white paper beneath.
- Place a few grains of two solids of citric acid and sodium carbonate on either side.
- Using a pipette make a puddle of water about 2cm in diameter, between the grains
- Use a wooden spill from either end move some grains into each side of the puddle.
- After a few minutes a line of bubbles appear (carbon dioxide). (See photo below left.
But I have got ahead of myself here. How can the particle theory explain the formation of silver iodide, when I have used particles of potassium iodide and silver or lead nitrate? How can silver and copper change places in the displacement reaction. How can these particles affect the colour of indicator dyes and cause bubbles of gas to appear. The particle model will have to be examined more closely and improved. New ideas will have to be introduced, but any refinements made will still have to explain the effects in this blog.
By starting with the solid
Chemistry is not magic. By moving our procedure one stage back, starting with the solid, the evidence is there that chemicals have entered the water, split apart (dissolved), moved (diffused) though the water. When two different chemicals meet they may (not always) react and solids, colour changes, or bubbles appear.
When thermal imaging becomes less expensive, I wonder if the reaction line will be seen. I have certainly seen it when the solids dissolve.
And we have saved time in preparation of solutions and disposal is just a wipe away with a paper towel.
The Johnstone triangle can be adapted to summarise these events which are met the everyday lives of students.