Chemistry is difficult to teach. You have frustrating moments when you want to hit your head against the wall and go home saying “is it because of me, they don’t understand?”
I picked up an old book of mine published 1977. One Chapter starts “You are perhaps puzzled why is a free energy change, rather than the enthalpy change, which is a measure of the ability of a reaction to do work.” Wonderful! Here is an author sympathising with the student (and teacher) that our subject is hard. Thankfully, I am not alone.
Those words were written by Alex H Johnstone and G Webb in the book, “Energy, Chaos, and Chemical Change”. Nobody writes these types of books now. At 110 pages, aimed at the teacher and the 6th form to undergraduate level, it just not cost effective for publishers. You would find this sort of material on the web nowadays. We dash things off in a couple of days (well some people do). I suspect this book took several months of care. It also requires careful but provides rewarding reading. The words are simple but the concept is deep. I often had (and still do) to go back to it because thermodynamics in chemistry is difficult.
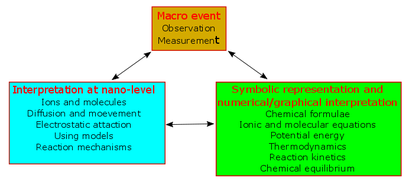
I was at a meeting of 40 chemistry teachers, having been invited to do a microscale workshop, with Professor David Read. David asked the teachers “How many of you have heard of Johnstone’s triangle”. (My version is on the right.) David is modern, he has electronic measuring equipment and they had to press a button, so we saw the answer immediately. Only one teacher had heard of it. When he put the triangle on the board, you could see flashing light bulbs coming from 39 heads. In this simple diagram, Alex Johnstone encapsulated the issues of teaching chemistry.
I picked up an old book of mine published 1977. One Chapter starts “You are perhaps puzzled why is a free energy change, rather than the enthalpy change, which is a measure of the ability of a reaction to do work.” Wonderful! Here is an author sympathising with the student (and teacher) that our subject is hard. Thankfully, I am not alone.
Those words were written by Alex H Johnstone and G Webb in the book, “Energy, Chaos, and Chemical Change”. Nobody writes these types of books now. At 110 pages, aimed at the teacher and the 6th form to undergraduate level, it just not cost effective for publishers. You would find this sort of material on the web nowadays. We dash things off in a couple of days (well some people do). I suspect this book took several months of care. It also requires careful but provides rewarding reading. The words are simple but the concept is deep. I often had (and still do) to go back to it because thermodynamics in chemistry is difficult.
I was at a meeting of 40 chemistry teachers, having been invited to do a microscale workshop, with Professor David Read. David asked the teachers “How many of you have heard of Johnstone’s triangle”. (My version is on the right.) David is modern, he has electronic measuring equipment and they had to press a button, so we saw the answer immediately. Only one teacher had heard of it. When he put the triangle on the board, you could see flashing light bulbs coming from 39 heads. In this simple diagram, Alex Johnstone encapsulated the issues of teaching chemistry.