Please do stand on my shoulders
I have been asked to contribute (amongst other contributions) to a Royal Society of Chemistry book on Creative Chemistry. I jotted down some thoughts and sent them back. It has been edited well but I thought it might be useful to add more here. is there such a thing as "Creativity" in school chemistry?
In school staff rooms the “Arts Teachers” claim (in a friendly way I find) that their subjects are more "Creative" because you need to express yourselves with originality. In science, they say, answers are just right and wrong. (I wish they were sometimes.) I understand what they mean but even the great painters, authors, poets, actors etc, have “stood on the shoulders of giants”, just as Isaac Newton, Albert Einstein etc have done.
Creativity is how you use techniques and discoveries made in previous generations, either in painting, poetry to explore and explain ones innermost feelings or in chemistry and electrical engineering to solve new problems. In science education, teachers need to be creative to allow students to understand the world around them, especially connections between various fields of science and engineering (and also economics). I have always stressed that teachers should create their own schemes of work and not rely on course books. Every school, every class is different.
Creativity is how you use techniques and discoveries made in previous generations, either in painting, poetry to explore and explain ones innermost feelings or in chemistry and electrical engineering to solve new problems. In science education, teachers need to be creative to allow students to understand the world around them, especially connections between various fields of science and engineering (and also economics). I have always stressed that teachers should create their own schemes of work and not rely on course books. Every school, every class is different.
Practical Work
Where practical chemistry work is possible in schools (such as the UK, students are often using the same procedures as their parents and possibly, even their grandparents did in school chemstry. Even if the presentation of lessons vary as in flipped, controlled investigation, sage on the stage, guide on the side etc, when it comes to procedure, methods hardly ever change.
Some of our standard practical procedures are up to 140 years old, can be found in old text books and are repeated in updated editions. Authors may be working from home with no access to the lab and in any case, practical procedure research can be very time consuming.
Continual repetition of these procedures takes no account of the changes in curriculum, examination specifications, education management, new equipment and materials, changes in safety legislation, environmental concerns, practical activities in university chemical education and information from research in chemistry education.
Some of our standard practical procedures are up to 140 years old, can be found in old text books and are repeated in updated editions. Authors may be working from home with no access to the lab and in any case, practical procedure research can be very time consuming.
Continual repetition of these procedures takes no account of the changes in curriculum, examination specifications, education management, new equipment and materials, changes in safety legislation, environmental concerns, practical activities in university chemical education and information from research in chemistry education.
Microscale chemistry procedures

These were developed in the USA to address green and waste issues and in South Africa to spread practical work, via UNESCO, to countries with limited resources. The kit was given to all UK schools by the RSC in 1999 along with a practical book. There were a few pockets of interest, but not many. It looked alien to eyes of many teachers at that time.
The involvement of CLEAPSS
Many teachers and academics think that CLEAPSS in the UK (http://science.cleapss.org.uk/ ) is just a safety police; we go around saying "don't do that!”. However, safety is only part of the remit. CLEAPSS development work centres on enabling teachers and technicians to deliver enjoyable, interesting activities which are safe (and legal) to perform. The Practical Procedures (PPdocs) are on the website for subscribers. These are also presented to teachers and technicians via courses run in England, Wales and Northern Ireland with SSERC doing much the same in Scotland. The revision of a procedure or new experiment should,
The developed microscale experiments have been presented in CLEAPSS safety talks, workshops and at ASE conferences. Visitors to CLEAPSS are shown the techniques in our laboratory. With interest finally shown from our examination boards, the techniques have become more popular.
The enthusiasts then began to report unexpected benefits.
- address activities that have caused safety issues such as toxic gas release, burns, explosions, etc,
- reduce the cost of expensive equipment by an alternative design or modern materials,
- address issues raised by UK safety legislation, usually designed for industrial purposes, but legally applying to school practical work,
- speed up practical work so that classroom management is more efficient,
- be directed to clarify an aspect of chemistry which causes confusion in students (misconceptions) and
- try to reduce time and effort in preparation and waste procedures.
- they would hamper students in answering questions exam questions based on older methods,
- they are not the exam-"required" method, even though the new method is more efficeint and more successful; also there is no requirement to do the published method, practical skills are required, and
- statements about microscale procedures such as “it is not proper chemistry”, “how can you do quantitative work”, “we have the proper equipment, why change?”, “it is for poorer countries” and “how can you demonstrate microscale chemistry?”
The developed microscale experiments have been presented in CLEAPSS safety talks, workshops and at ASE conferences. Visitors to CLEAPSS are shown the techniques in our laboratory. With interest finally shown from our examination boards, the techniques have become more popular.
The enthusiasts then began to report unexpected benefits.
- Lab room management was less stressful..
- Students were more focussed and not side-tracked by other student groups.
- Mistakes were quickly and safely corrected.
- The practical sessions were completed in the time available..
- The results were visually pleasing, could be photographed and inserted into the lab book for future reference.
- The use of webcams, visualizers and USB microscope allow dramatic demonstrations.
- There was a reduction in the overload of the short-term working memory and so students were not asking “what do I do next?”
- The practical work could be focussed on addressing known misconceptions and difficult areas in chemical education.
- Completely new and interesting experiments could be developed
Creativity?
Sudden inspirations and “eureka” moments do happen when an idea comes together but there are many failures along the way. I have over sixty years of experience in school practical work (man and boy). I started looking for alternative procedures as early as 1993 for CLEAPSS and it was not for another 10 years that I realised I was creating something new and accessible. All the time I was building on the work from other sources such as Nuffield Chemistry, Education in Chemistry, Journal of Chemical Education and School Science Review (Association of Science Education) and various sources on the web with the Bruce Mattson Microscale Gas Chemistry really acting as a catalyst. Also from from like-minded people in other countries such as the USA, Germany, China, Japan, South Africa, Thailand and Malaysia who meet every two years.
There is added creativity in adjusting or modifying what occurs in these countries to the chemistry required in the England GCSE and A level. SSERC (in Scotland) have also developed microscale techniques alongside those from CLEAPSS. Here are some examples.
There is added creativity in adjusting or modifying what occurs in these countries to the chemistry required in the England GCSE and A level. SSERC (in Scotland) have also developed microscale techniques alongside those from CLEAPSS. Here are some examples.
Reactions in Puddles

This is only made possible by using plastic folders or laminated worksheets. The idea came from the RSC book on microscale chemistry but they used Overhead Transparency Sheets. The advantage with folders and lamination is that instructions can be asily seen and followed.
A puddle of water is about 1.5cm diameter. The volume of liquid is less than 0.8cm³. The picture shows the the colours of buffers with different indicators and then combining the indicators to prepare a Universal Indicator
Displacement and precipitation reactions can be investigated. Basically, the test tube is replaced by the puddle.
Solutions can be used but it is a better teaching point to push the solid directly or introduced it on the end of damp pointed sticks,letting the solid dissolve and the solvated ions diffuse. See video below.
This resulted in a paper (and front cover) of the the May 2019 edtion of the Journal of Chemical Education.
(Bob Worley, Eric M. Villa, Jess M. Gunn & Bruce Mattson* J. Chem. Educ. 2019, 96, 5, 951-954)
A puddle of water is about 1.5cm diameter. The volume of liquid is less than 0.8cm³. The picture shows the the colours of buffers with different indicators and then combining the indicators to prepare a Universal Indicator
Displacement and precipitation reactions can be investigated. Basically, the test tube is replaced by the puddle.
Solutions can be used but it is a better teaching point to push the solid directly or introduced it on the end of damp pointed sticks,letting the solid dissolve and the solvated ions diffuse. See video below.
This resulted in a paper (and front cover) of the the May 2019 edtion of the Journal of Chemical Education.
(Bob Worley, Eric M. Villa, Jess M. Gunn & Bruce Mattson* J. Chem. Educ. 2019, 96, 5, 951-954)
Using Diffusion to Transfer Chemicals
Chemicals contain small invisible particles in random motion. We can see the results of any interaction between these particles in chemical reactions by changes of colour, solids appearing, changes in temperature etc. Diffusion can only be explained by particle theory but is introduced very early in the UK curriculum but then hardly reappears. In fact, some modern syllabuses do not mention “diffusion” at all. I can see that it is implied in other specifications. Possibly, diffusion appears so obvious to teachers/examiners that we miss it out. Yet research shows it is a very difficult concept for students to understand..

Based on original ideas in China and the RSC book, the use of small Petri dishes (never previously used in chemistry and quite alien to teachers with a traditional outlook) allowed the use of puddles with diffusion. The normal scale electrolysis of copper and sodium chlroide had exposed students to chlorine and they had been taken to hospital with breathing difficulties. Microelectrolysis of 0.5M copper chloride solution only produces around 6 cm³ of chlorine gas and yet, this is enough to carry out reactions on damp blue litmus and “puddles” of 0.1M potassium iodide solution and 0.5M potassium bromide solution. Finding more robust electrodes of 2mm medium wall carbon–fibre rods improved the procedure.
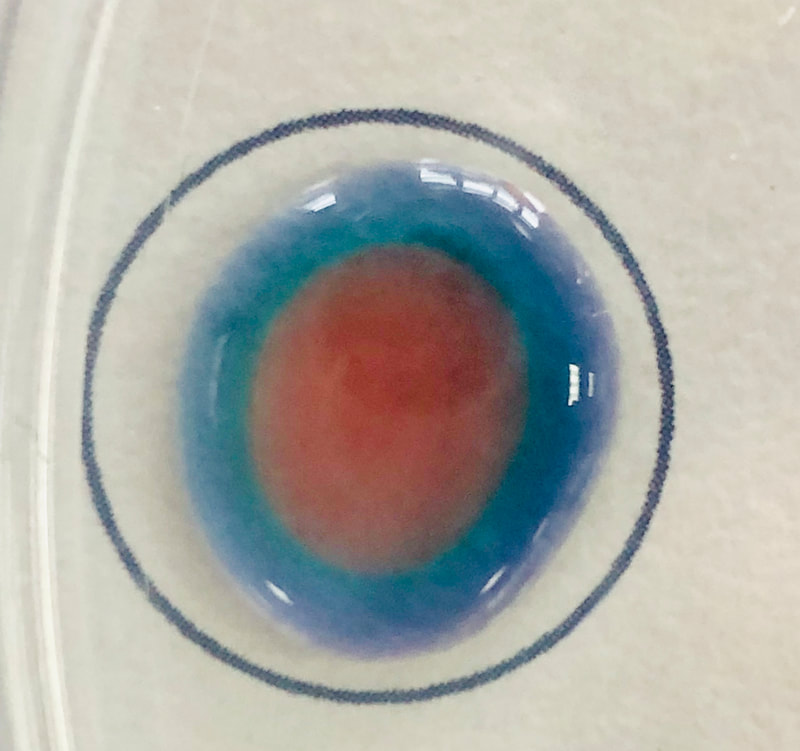
I then generated gases in Petri dishes chemically and produced amazing diffusion halos as in the example below with ammonia molecules in the Petri dish diffusing into a puddle of 0.1M hydrochloric acid with Universal Indicator. This is followed by hydroxide ions diffusing though the liquid, neutralizing the acid. These prcedures only take a few minutes.
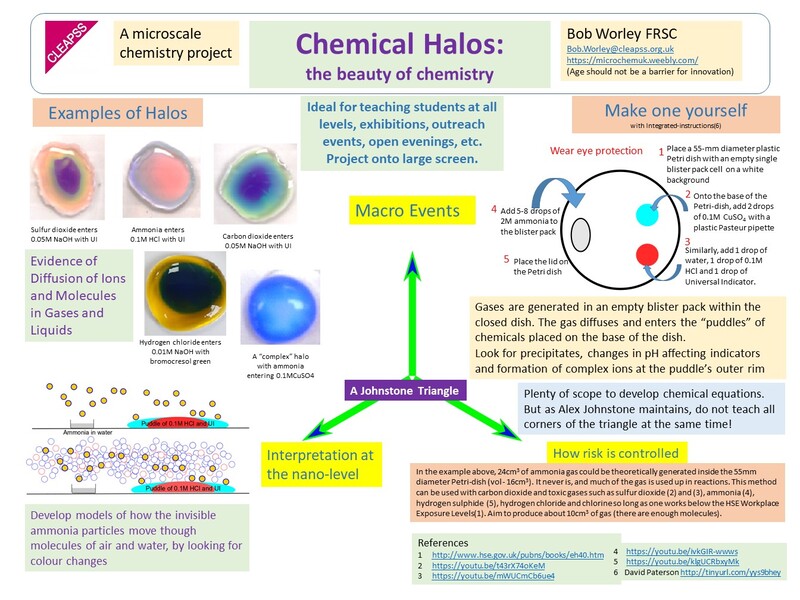
So it has been quite a year with the chemcial halo poster winning the RSC Twitter Poster Competition for Further Education. AS you can see in the poster, other halos are avialable.
I then generated gases in Petri dishes chemically and produced amazing diffusion halos as in the example below with ammonia molecules in the Petri dish diffusing into a puddle of 0.1M hydrochloric acid with Universal Indicator. This is followed by hydroxide ions diffusing though the liquid, neutralizing the acid. These prcedures only take a few minutes.
So it has been quite a year with the chemcial halo poster winning the RSC Twitter Poster Competition for Further Education. AS you can see in the poster, other halos are avialable.
“Where practical chemistry work is not possible in schools”
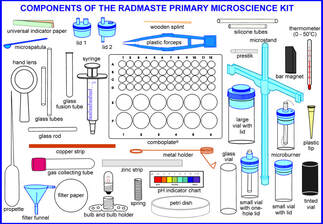
My efforts in creativity pale into insignificance when compared to that of John Bradley in inventing the Radmaste kits, promoted by UNESCO. Yes, they have been used worldwide but they have come up against the same barrier as microsale in the UK, Traditiionally trained curriculum advisers and exam setters do not accept the changes. The initial enthusiastic users of the kits come up against lack of finance in maintainance of the kits and continued professional development..
Is creativity in practical procedures, seen as a part of research in chemical education?
I sometimes wonder about this although I am assured by many that it is a part of chemcial education research. However, barriers are put in the way of presenters when trying to present these procedures at important international meetings.
- The venue is not a University but a hotel (Hotel safety officer objects!).
- The venue is not in the chemistry department of the University. (University safety officer objects)
- The chemistry department is a some distance from the venue.
- Safety is run on Hazard Assessment as opposed to Risk Assessment. (Chemistry safety oficer does not understand microscale techniques)
- A University risk assessment proforma, designed for research is not applicable to this type of chemistry.
- Sorry to say but many of those who lead chemical education research from various countries are visible by their absence from practical workshops, by not taking part and not even visiting them.
- How can you see microscale? Well most venues have excellent IT facilities such as visualizers and split screens upon which the experiments can be displayed. So lecture demonstrations are possible.
- Journals demand evidence of the effectiveness of the work on students as well as a direct technical account of a new procedure. I can see that this is jumping through hoops in many cases. I prefer to let teachers see the results on Twitter and You tube. It is more immediate and effective than writing in a peer-reviewed journal, which teachers cannot easily access anyway. When teachers see these experiments or used them in workshops, light bulbs come on in their minds.