I read in Education in Chemistry from Kristy Turner and Nathan Owston that many students draw the curly arrow coming from hydrogen ions or positive (electron deficient) species rather than a particle with a lone pair of electrodes. There could be two factors here, one chemical and anthropomorphic,
- our reliance on the Lowery-Bronsted and Arrhenius definitions of acids and bases in our courses to the detriment of the Lewis definition and
- a deep-seated love of the “positive (good)” and wariness of the “negative (bad)”, a sort of hidden-anthropomorphic effect which challenges our common sense.
Acid and Bases
In the table below, there are the 3 definitions of acids and bases.

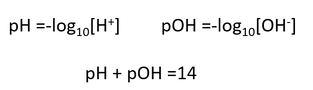
The definition of acid for students up to 16 years old is the Arrhenius definition. This leads to a scale of acidity, known as the pH scale of 0-14. Why is there not a scale for alkalinity? There is one course, the pOH scale.
| The scale for alkalis, pOH, is seldom used. In one Board’s syllabus, the pH term is mentioned 14 times at GCSE but pOH is not mentioned either at GCSE or even at A level. In practical work, students use pH indicator paper but what about pOH values. We might use pH meters but the values are in pH and never in pOH. Commercial pOH meters are not even available! Everything students learn is geared towards the positive hydrogen ion and not the negative hydroxide ion. It is almost as through the student can dismiss the negative ions. | |

The emphasis on acid is not surprising; acids react with metals, hydroxides and carbonates in a reasonably straightforward manner; not so with alkalis, which have a complicated reaction with metals such as zinc and aluminium. At GCSE the reactions of alkalis is limited to the reaction with hydrogen ions to form water.
By the time the student has completed this section of education, there is more knowledge about the positive ions than the negative ions. There is also that underlying hidden-anthropomorphic urge to think that it is "positive" the student should be concentrating on not the “negative”.
By the time the student has completed this section of education, there is more knowledge about the positive ions than the negative ions. There is also that underlying hidden-anthropomorphic urge to think that it is "positive" the student should be concentrating on not the “negative”.
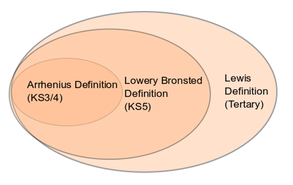
The same Awarding Body introduces the Lowry-Bronsted definition in the next stage of chemistry. It is there because a wider definition of acids is required to cope with reactions where water is not involved, eg, dry ammonia reacting with dry hydrogen chloride to form solid ammonium chloride. It is also required in the discussion of equilibria in weak acids and alkalis. The diagram on the right shows the linear view which we are forced to adopt. Up to now, there is no direct mention the Lewis definition at school level so it will be introduced a the next stage of education, although it is the Lewis definition which links acid/base definitions with reaction mechanisms.
"Curly arrows"
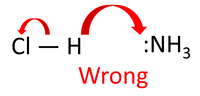
When a hydrogen chloride (HCl) molecule collides with an ammonia molecule (NH₃) in the right orientation, a student, brought up thinking that acids, hydrogen ions acting as a proton donor. Would try to write this as a mechanism. Of course, there is an issue in how the chloride ion forms.
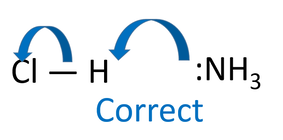
If the students are taught the Lewis definition and use the electron donor definition then the curly arrow mechanism just flows. Ammonia is a Lewis Base, donating electrons to the hydrogen atom (δ+vE). This causes hydrogen chloride bond to break so there is the chloride anion and ammonium cation. chloride ion. Ammonia is called a nucleophile in organic chemistry. Is a Lewis Base and a Nucleophile the same thing?
87t
Keeler and Wothers in “Chemical Structure and Reactivity” write “there is little distinction between the terms electrophile and Lewis acid (or nucleophile and Lewis base) but the latter is used when the species in question forms and adduct (which could be isolated as an intermediate)”.
87t
Keeler and Wothers in “Chemical Structure and Reactivity” write “there is little distinction between the terms electrophile and Lewis acid (or nucleophile and Lewis base) but the latter is used when the species in question forms and adduct (which could be isolated as an intermediate)”.
Why should the mechanism show direction from the negative species (the Lewis Base)?
Peter Atkins writes
Peter Atkins writes
The statement “An electron falls into a hole” was a "lightbulb" moment for me. Reactions should be viewed as electron-rich species donating its electrons to an electron poor species. But if students are not taught the Lewis definition, they will not see the connection.
There are separate contexts for the definitions of acid and base.
There are separate contexts for the definitions of acid and base.
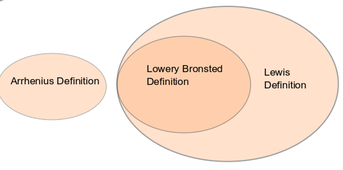
- Arrhenius definition describes the extent of acidity of alkalinity at a certain time.
- The Lewis Definition (the Lowery-Brønsted definition being inclusive) defines a process or reaction pathway of electron donation and acceptance. This has a close connection to reaction mechanisms and to “curly-arrow” chemistry).
My conclusions are that
- pOH should be taught alongside pH to students to GCSE level.
- The Lewis definition of acids and bases should be covered at A level, to include the Lowry/Brønsted definition.
“But the syllabus is too full as it is.” Will come the cry. The whole of chemistry is intertwined. However, we still tend to teach the subejct in three separate parts, Physical, Inorganic and Organic. In teaching the Lewis definition we are connecting Reaction mechanisms (Organic) with the theory of acids and bases (Physical). The more connections one makes in chemistry the more light-bulb moments your get.
Then there is that positive is “good”, negative is “bad” thought niggling away in the back of our minds. It might be true in life that we should always look at the positive aspects of living (the bright side of life). Not in chemistry though; there is always a balance between positive and negative. There is a lovely song written in 1944 begins with “You’ve got to accentuate the positive, eliminate the negative”. Here the Jules Holland version.
