My microscale activities have to add something extra that traditional procedures cannot normally achieve. This can range from ease of presentation, better classroom management to, what I consider the most important, provide further evidence to students in how chemicals interact to form new substances.
On twitter, these little videos and photos can circle the world from London to Phoenix, Auckland, Sydney, Hong Kong, Ankara, Salzburg to Bolton back in the UK. I do not get many hits on the whole and if I reach 1000, it is very gratifying but two videos seemed to hit the spot recently. The video with the formation of the red slash of the iron-thiocyanate complex has achieved about 4,500 media engagements (MEs).
On twitter, these little videos and photos can circle the world from London to Phoenix, Auckland, Sydney, Hong Kong, Ankara, Salzburg to Bolton back in the UK. I do not get many hits on the whole and if I reach 1000, it is very gratifying but two videos seemed to hit the spot recently. The video with the formation of the red slash of the iron-thiocyanate complex has achieved about 4,500 media engagements (MEs).

- However, the video with the red slash being deformed when carried out over a strong magnet received only 918 MEs. See https://www.youtube.com/watch?v=mS1o9Dn7nRM. The appearance of red slash is must be more visually appealing than my ablility to bend it. Perhaps people think "this is going to be diffiult to explain, I don't think I will bother" but here is evidence of paramagentism. It is not an easy subject and viewers wil be swticed off straightaway.
It is caused by the number of unpaired electrons in the iron complex. To me it was wonderful, to the many it was not. I realsied I was a nerd!
| A video of the chemistry of sulfur dioxide received 650 MEs but the follow up video which described the chemicals involved gets 63. As most of my subcribers are chemists and teachers of chemistry, you would have thought they want to know what was what, especially the colourless paper which goes blue and then colourless again – an indicator paper new to school science. Potassium iodate/starch paper is recommended as a greener test for sulfur dioxide gas above potassium dichromate. | |
The video with the greatest number of MEs involves placing sodium sulfate in water with Universal Indicator (UI) above a strong magnet and then insert carbon-fibre electrodes and applying a DC current. There have been over 8500 MEs. The circulating colours are indeed wonderful to watch but is there an educational use for this experiment? The only that has going for it is that it shows the effect of a magnet on ions, possibly similar to that in a Mass Spectrometer.
| The much more relevant video for teaching where a purple potassium manganate(VII) crystal is inserted into a puddle with a magnet below and electrodes applied has had only 732 views. With the purple colour circulating around the positive electrode, this is really evidence for a negatively charged manganate(VII) ion. | |
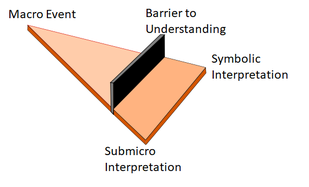
So what do infer from this. Well much is written about Alex Johnstone triangle and cognitive overload applying to students. I reckon it also applies to teachers as well. We all love the macro events but wanting to know more and how they can lead to a greater understanding of chemical interaction at the submicro (nano) level and to understand the symbolic and mathematical nature requires to the students AND the teachers to overcome a barrierof understanding. It is rather like the activation energy in a chemical reaction. The teacher is crucial here because, rather like acting as a catalyst, the teacher can reduce the height of the barrier though good teaching..
Going beyond the spectacular is a difficult process which we, as teachers, realise. If we can recognise the macro barrier in ourselves then we become better teachers.
Going beyond the spectacular is a difficult process which we, as teachers, realise. If we can recognise the macro barrier in ourselves then we become better teachers.