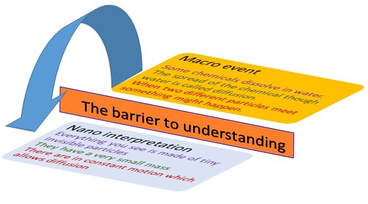
Mounting the barrier of understanding between the macro event in a chemistry event and the interpretation at the nano level is one the most difficult achievements for both the student and the teacher..
Where does the chemistry teacher start with chemistry? The typical traditional start in UK schools was with the destructive nature of the Bunsen burner with safety to introduce practical work and then onto elements, mixtures and compounds (which is quite a difficult concept) with separation techniques.
Where does the chemistry teacher start with chemistry? The typical traditional start in UK schools was with the destructive nature of the Bunsen burner with safety to introduce practical work and then onto elements, mixtures and compounds (which is quite a difficult concept) with separation techniques.