Reading some authors, you would think a “WOW” moment in chemistry at the age of 14 or so, is an event that makes such a massive impact in the way you feel about taking the chemistry A level exam per year, is this good evidence for such a claim? They also usually choose chemistry as a prerequisite for other studies, eg medicine. I was fortunate if I had 2 students (per hundred cohort) moving on to read chemistry at university.
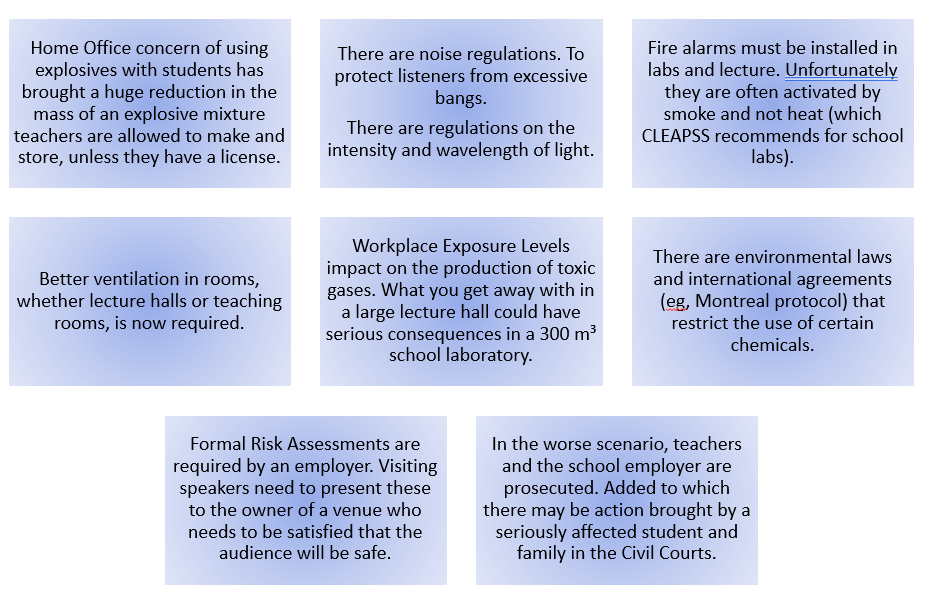
Chemistry teachers are fortunate to have at hand some eye-catching demonstrations which they can parade in front of students. However, we are also persuaded (by school managers) to exhibit these “wow” moments during open evenings to inspire new students and their parents and to spark interest in primary school students visiting a school the year before they attend. There have been some unfortunate incidents in these events usually caused by poor procedure. Explosions and fire have been known to scare younger students and upset adult visitors alike. (Our work at CLEAPSS involves investigating these incidents and from our findings, develop new and improved control measures to make procedures safer but not reduce any impact of the demonstration. CLEAPSS does not ban experiments but interpretation of UK safety and environmental legislation has made big differences to many of the older methods.
Chemistry teachers are fortunate to have at hand some eye-catching demonstrations which they can parade in front of students. However, we are also persuaded (by school managers) to exhibit these “wow” moments during open evenings to inspire new students and their parents and to spark interest in primary school students visiting a school the year before they attend. There have been some unfortunate incidents in these events usually caused by poor procedure. Explosions and fire have been known to scare younger students and upset adult visitors alike. (Our work at CLEAPSS involves investigating these incidents and from our findings, develop new and improved control measures to make procedures safer but not reduce any impact of the demonstration. CLEAPSS does not ban experiments but interpretation of UK safety and environmental legislation has made big differences to many of the older methods.
Some legal requirements on “Wows”
Learning from past incidents
UK Law places the responsibility of safety at work on the employer (usually a non-scientist). A teacher is an employee. A student is a visitor. The teacher has to carry out risk assessment on behalf of his employer and follow control measures in any “wow” experiment to ensure bot employees and visitors are safe. My organization, on working on behalf of their employer, assists teachers in this procedure but incidents still happen. A Whoosh bottle explodes rather than goes “Whoosh”. An explosive vapour/air mixture has formed in the bottle because of a delay in igniting the contents, once the bottle had been primed. Hence, we note in our risk assessment, the control measure that teachers “Do not delay the ignition process. (This usually happens to save preparation time or a long introduction) Issues have arisen if bottles are left to stand before ignition”.
Issuing trigger warnings
carelessly Loud bangs, fire, ultra-cold liquid gases, extremely bright light, corrosive liquids, nasty smells, slimy liquids are fascinating but some people are scared due to past traumas. Teachers do not know what traumatic events might have occurred a child’s past. “Hands over ears” for a hydrogen oxygen balloon in a Hall or dynamite soap bubbles in a lab. Some would call this “woke” but what if a child has recently been at a funeral of a grandparent or worse still, a sibling, and the teacher talks about cremating a jelly baby?
Training/Rehearsal
Consummate performers who carry out demos as a profession, do loads of rehearsal. Demonstrators range from the “sage on stage” to the bumbling clown, via the whacky professor (with crazy hair and bow tie!) Have you seen how adult spectators over-react to chemistry demos on TV? (Please do not get me started on the depiction of mad scientists in chemistry labs in TV and film dramas!). Teachers are not at school to entertain but to teach; by all means, make the experience enjoyable and they must rehearse the procedure. And as one Irish comedian used to say "It's the way you tell 'em. Chemistry is not magic but you can learn a lot from stage magicians how to build up tension in the audience.
Audience Suitability
Does a seven year old really understand that a gas can exist as a liquid at -196°C? That a tennis ball at this temperature does not bounce but splinters into many pieces. Others will like the white fumes of dry ice in hot water, because that looks like “science” on films and TV. There is no harm in this so long as presenters do not throw liquid nitrogen over the audience, pour it along the floor with children seated a few yards away or reduce the oxygen content of the room. Other than looking like a firework, what does a thermit reaction show an eleven-year old?
A chemical demonstration should not appear as magic, a sleight of hand; in education there should be a rational explanation that makes sense of what happens to the audience. A liquid suddenly changes from colourless to black because it is a clock reaction. In teaching, a students should learn from applying their senses to a demonstration not just be entertained.
A chemical demonstration should not appear as magic, a sleight of hand; in education there should be a rational explanation that makes sense of what happens to the audience. A liquid suddenly changes from colourless to black because it is a clock reaction. In teaching, a students should learn from applying their senses to a demonstration not just be entertained.
“Wows” and our senses
There are more than 5 senses but the these are the ones by which students experience an event. With touch and tasting experiences, the demonstrator has to be very careful;
Experience |
Example |
Comment |
Explosion (hearing) |
From squeaky pops to hydrogen oxygen explosions. |
But beware: Students may be deaf and need to adjust hearing aids. They can scare students. They can perforate ear drums and blow out windows. Transferring a demonstration in a lecture to a small lab (300m3) is a part of the risk assessment. |
Fire, sparks (sight and hearing) |
From burning magnesium to Whoosh Bottles and Howling Jelly Babies |
But beware: They can scare students. The light may be in the UV area. Particles can shoot out; objects can catch fire. Fire alarms can be set off. |
Sudden Colour changes (sight) |
Precipitates, indicators, reaction kinetics |
But beware: Do some students have sight issues and be colour blind? |
Smell (Olfactory) |
Odours from ammonia, hydrogen sulfide, chlorine, vinegar, onions, esters, organic chemicals |
But beware: detecting odours means the chemicals have entered the human body through the lings. There are Workplace exposure levels to deal to contend with. |
(Touch) |
Solids and liquids. Slime. Hot or cold? |
But beware: some liquids might be corrosive! |
(Taste) |
Food only |
But beware: No tasting in the laboratories. |
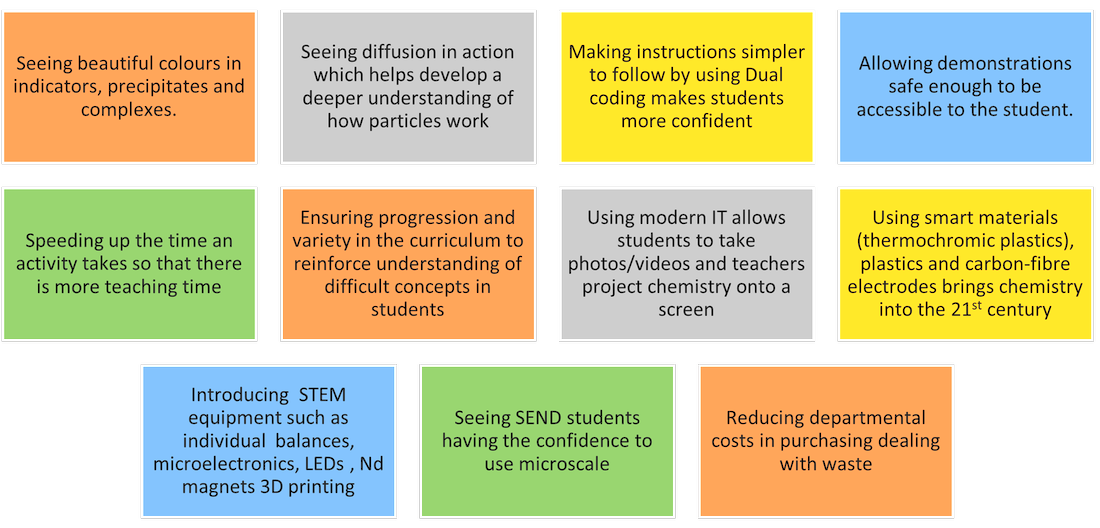
Our Achievement Wows
The students (and teachers) are experiencing chemistry in day to day life, from washing their hands and face (surfactants and precipitates) to using their mobile phones (over sixty different elements being used.). When the teacher points this out, we might get a “micro-wow” reaction. I often prefer a series of “micro-wows” rather than one big one. I can see this in adults when I lead them through a series of microchemistry activities in a workshop. (It brings me great satisfaction!).
Experience |
Comment |
“Wow, you mean that’s chemistry?” |
Teachers can relate the chemistry taught to an occurrence that students meet regularly in their day-to-day lives from having a wash (surfactants and precipitates) to using their mobile phone (over sixty different elements being used). What chemistry is there in a car? What chemicals are they wearing from materials to make up? What chemicals are they eating and drinking? What elements are inside their bodies. |
“Wow, I never knew that, amazing!“. |
If the am necueasu is a cirle with I did not realise there so much empty space in an atom”. There are more molecules in 18 g of water than there are stars in the universe |
“Oh I see, Wow”. The penny drops |
Balancing an equation, drawing graphs form own data, performing a calculation correctly, can all bring that “eureka” moment. Then comes an explanation of a chemical processes by means of ions and molecules (invisible particles) or through energy (an abstract concept), which show that an understating of the subject is beginning. |
“Wow, look what I’ve done” |
Students taking pride in managing a mechanical skill; Chemistry can be just sloshing liquids around, in a devil-may care fashion to look nonchalant, cool, etc. In fact there are teachers who enjoy this approach, despite spilling liquids etc. However, chemistry requires more careful skills such as adding a drop form a burette, syringe or pipette, setting up equipment, taking data or bending glass tubing in a Bunsen flame. Why not present achievement certificates? |
“Wow! I have never seen that before”. |
This happens all the time when carrying out small scale experiments. Recently I have established that that ozone is made as a secondary reaction during electrolysis in a Hofmann voltameter. Even though the concentration is small (0.2%) it certainly makes dynamite soap louder! |
Can microchemistry and sustainable chemistry have “wow” moments?
I am told that micro and small-scale chemistry, cannot have “wow” moment. On a personal level I am always astounded at what I see close up,
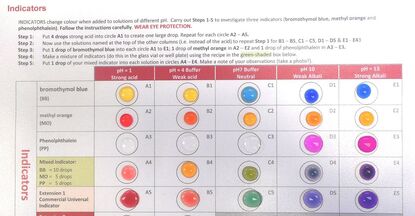
My introductory experiment to adults is making a universal indicator. This is all done on a polypropylene folder with the worksheet inside. There are 25 reactions. Teachers see how a Universal indicator works and Technicians see there is no washing up of test tubes afterwards and both are amazed how beautifully it is presented on the plastic surface: a triple micro-wow make a big “wow”! . As an introduction for students, teachers could extend the method to discovery with natural products from the garden. Every flowerhead, coloured leaf or fruit skin is a universal indicator. Everyone knows about red cabbage but try butterfly-pea tea, dark-coloured petunias or the coloured oxalis or tradescantia leaves.
My introductory experiment to adults is making a universal indicator. This is all done on a polypropylene folder with the worksheet inside. There are 25 reactions. Teachers see how a Universal indicator works and Technicians see there is no washing up of test tubes afterwards and both are amazed how beautifully it is presented on the plastic surface: a triple micro-wow make a big “wow”! . As an introduction for students, teachers could extend the method to discovery with natural products from the garden. Every flowerhead, coloured leaf or fruit skin is a universal indicator. Everyone knows about red cabbage but try butterfly-pea tea, dark-coloured petunias or the coloured oxalis or tradescantia leaves.

Add magnesium to iron(II) sulfate sol and you see iron forming on the metal (which makes it attracted to a magnet (red object in the right of the photo)) but there is a consecutive reaction as bubbles of gas (hydrogen) and a green precipitate appear changing to brown as air oxidizes iron(II) hydroxide to iron(III) hydroxide. Iron acts as a catalyst on the surface of magnesium to increase the rate of the magnesium/water reaction to produce the hydrogen and iron(II) hydroxide.
“Is this supposed to happen?” is the wonderful question from students. As though particles enjoy carrying out reactions with the sole purpose to mislead students. The challenge is how to interpret these effects using the invisible particles (ions and molecules) in action. It will challenge teachers because results are not always as the textbook/specification suggests they should be.
“Is this supposed to happen?” is the wonderful question from students. As though particles enjoy carrying out reactions with the sole purpose to mislead students. The challenge is how to interpret these effects using the invisible particles (ions and molecules) in action. It will challenge teachers because results are not always as the textbook/specification suggests they should be.
Am I over complicating the subject? Consecutive and secondary reactions often occur alongide the main reacionin chemistry. The authors of text books hardly ever mention them. What we do in these reactions is not to stir them but to let the particles do the work and then we get many more micro-wows as we observe the results of random motion and collisions of invisible particles. We like to give the impression that Chemistry is a right and wrong subject. It is not. It is fuzzy. These micro-wows (see below) enthuse not only the student but the teaching staff as well.
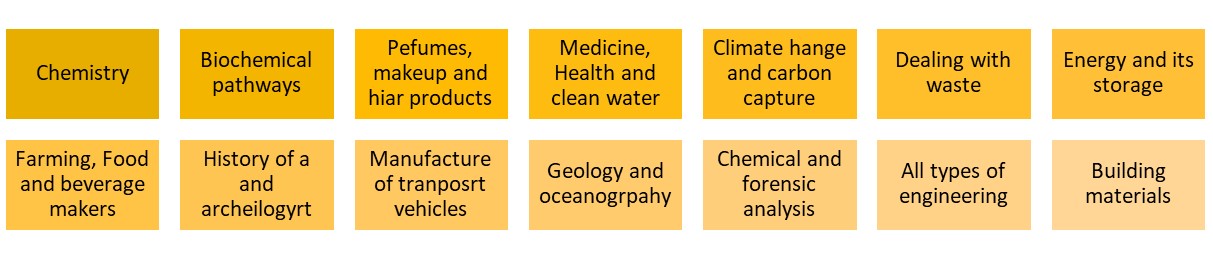
Wows from connecting chemistry with other disciples
| When the questions comes “what use is chemistry to me if I become a ………?” Chemistry has tentacles in other disciplines and areas of work. Examples are in the yellow yellow boxes below. There are many more such as education itself. In the course of teaching they can be used in teaching as examples of the uses of chemistry. In the History of art, chemical analysis is used to date paintings, as some pigments were not available until a certain date. Now what are the reasons for balancing chemical chemical equations? Chemistry is not magic. We cannot make matter but can alter it and the micro approach to the conservation of mass (see right) can easily show this. | |
The Holistic Hinterland
And with this comes a holistic approach to looking at chemistry through areas which reflect the complexity of life because of the interaction of the subject with non-chemists and society. This is called systems thinking and I used to cover this in courses such as Science in Society, a AO and AS level course for those in Y12 during the 70s and 80s. This can garner a response which I will call “anti-wow” when students and adults are so overwhelmed by the issues in the world, they turn their backs on science. I first saw this with the hippie movement in the 1960s cause by the impact of nuclear weapons on society and ease to which drugs such as LSD could be made and provided. Now there are active science deniers and conspiracy theorists commentating on vaccination, climate change etc.
On the other hand though:
Humans have the capacity to solve problems and provide answers. Examples such as such as wind farms, solar panels, batteries to drive cars, extract rare elements from ever decreasing supplies, make plastics from other sources, use oil in a responsible way etc, provide ant-viral drugs, many of which were unheard of 10 years ago.
On the other hand though:
Humans have the capacity to solve problems and provide answers. Examples such as such as wind farms, solar panels, batteries to drive cars, extract rare elements from ever decreasing supplies, make plastics from other sources, use oil in a responsible way etc, provide ant-viral drugs, many of which were unheard of 10 years ago.
But are “Wows” in Exam Questions?
Teachers often claim that the micro approach disadvantages the students because the procedures do not appear in exam questions. Well, nor are Whoosh bottles and Howling Jelly babies (combustion, redox), elephants toothpaste (rate of reaction, catalysis, decomposition, redox) and Thermite (redox, displacement) yet they are still shown by teachers. The ramifications of chemistry in society, (the hinterland) is vast. It invites the question from students ”do I need to know this for the exam?”.
Examinations answers usually hinge on correct-word answers or calculations but in real-life, the hinterland of chemistry is fuzzy and answers in the hinterland area require discussion through extended essay. These are not easy to mark.
Aspects of science may have caused these environmental and health issues. However, the answers lie in science but the moral, ethical, and economic will has to be there. as well and politicians have to bring it all together. As I write, the general public are having to be dragged kicking and screaming to update their cars, just as they were when smokeless fuels were introduced, we all had to wear safety belts in cars, told to stop smoking cigarettes, to follow lockdown procedures and have vaccinations etc.
Communicating chemistry to the general public is an artform in itself. The image is poor and we do not have a charismatic figure or National; Treasure such as Brian Cox or David Attenborough in our ranks, I do not think we do ourselves any favours by just relying on "WOW" experiences based on the senses alone in broadcasting and writing.
Examinations answers usually hinge on correct-word answers or calculations but in real-life, the hinterland of chemistry is fuzzy and answers in the hinterland area require discussion through extended essay. These are not easy to mark.
Aspects of science may have caused these environmental and health issues. However, the answers lie in science but the moral, ethical, and economic will has to be there. as well and politicians have to bring it all together. As I write, the general public are having to be dragged kicking and screaming to update their cars, just as they were when smokeless fuels were introduced, we all had to wear safety belts in cars, told to stop smoking cigarettes, to follow lockdown procedures and have vaccinations etc.
Communicating chemistry to the general public is an artform in itself. The image is poor and we do not have a charismatic figure or National; Treasure such as Brian Cox or David Attenborough in our ranks, I do not think we do ourselves any favours by just relying on "WOW" experiences based on the senses alone in broadcasting and writing.