Chemistry is difficult to teach. You have frustrating moments when you want to hit your head against the wall and go home saying “is it because of me, they don’t understand?”
I picked up an old book of mine published 1977. One Chapter starts “You are perhaps puzzled why is a free energy change, rather than the enthalpy change, which is a measure of the ability of a reaction to do work.” Wonderful! Here is an author sympathising with the student (and teacher) that our subject is hard. Thankfully, I am not alone.
Those words were written by Alex H Johnstone and G Webb in the book, “Energy, Chaos, and Chemical Change”. Nobody writes these types of books now. At 110 pages, aimed at the teacher and the 6th form to undergraduate level, it just not cost effective for publishers. You would find this sort of material on the web nowadays. We dash things off in a couple of days (well some people do). I suspect this book took several months of care. It also requires careful but provides rewarding reading. The words are simple but the concept is deep. I often had (and still do) to go back to it because thermodynamics in chemistry is difficult.
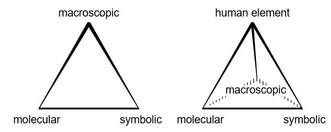
I was at a meeting of 40 chemistry teachers, having been invited to do a microscale workshop, with Professor David Read. David asked the teachers “How many of you have heard of Johnstone’s triangle”. (My version is on the right.) David is modern, he has electronic measuring equipment and they had to press a button, so we saw the answer immediately. Only one teacher had heard of it. When he put the triangle on the board, you could see flashing light bulbs coming from 39 heads. In this simple diagram, Alex Johnstone encapsulated the issues of teaching chemistry.
I picked up an old book of mine published 1977. One Chapter starts “You are perhaps puzzled why is a free energy change, rather than the enthalpy change, which is a measure of the ability of a reaction to do work.” Wonderful! Here is an author sympathising with the student (and teacher) that our subject is hard. Thankfully, I am not alone.
Those words were written by Alex H Johnstone and G Webb in the book, “Energy, Chaos, and Chemical Change”. Nobody writes these types of books now. At 110 pages, aimed at the teacher and the 6th form to undergraduate level, it just not cost effective for publishers. You would find this sort of material on the web nowadays. We dash things off in a couple of days (well some people do). I suspect this book took several months of care. It also requires careful but provides rewarding reading. The words are simple but the concept is deep. I often had (and still do) to go back to it because thermodynamics in chemistry is difficult.
I was at a meeting of 40 chemistry teachers, having been invited to do a microscale workshop, with Professor David Read. David asked the teachers “How many of you have heard of Johnstone’s triangle”. (My version is on the right.) David is modern, he has electronic measuring equipment and they had to press a button, so we saw the answer immediately. Only one teacher had heard of it. When he put the triangle on the board, you could see flashing light bulbs coming from 39 heads. In this simple diagram, Alex Johnstone encapsulated the issues of teaching chemistry.

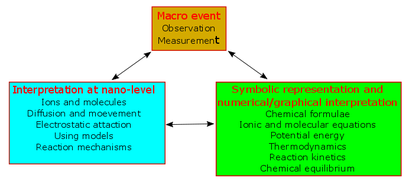
Alex knew the problems. Before entering Glasgow University, he taught in a secondary school in Scotland for 13 years. He wrote a chemistry syllabus book in 4 volumes. called “Chemistry Takes Shape”. This was published in 1965. I have copied out a small section of page 2 of the first volume. You can see his style all over it. In this section, he is teaching about small particles to the first year of Scottish High School Chemistry. Here are the macro events being interpreted at the nano-level from the very first student activities.
The Bunsen Burner can wait!
(See Appendix for a version you can try).
He also produced a textbook called Chemistry About Us, (Heinemann) in 1988. He wrote in 2000, "We were also lucky to publish at a time when the curriculum had not been set in concrete. Teachers were open to ideas. Now there is a real pressure for textbook writers to conform to the set concrete pattern of the “accepted” curriculum. Woe betide anyone who is too adventurous. His sales are doomed." He was so right.
The Bunsen Burner can wait!
(See Appendix for a version you can try).
He also produced a textbook called Chemistry About Us, (Heinemann) in 1988. He wrote in 2000, "We were also lucky to publish at a time when the curriculum had not been set in concrete. Teachers were open to ideas. Now there is a real pressure for textbook writers to conform to the set concrete pattern of the “accepted” curriculum. Woe betide anyone who is too adventurous. His sales are doomed." He was so right.
The triangle was my first light bulb moment from Alex Johnstone but there were more. If you put Alex H Johnstone into Google, you will find many paperswritten by him and his students (https://www.researchgate.net/profile/Alex_Johnstone) . I know of many teachers at the present time are much exercised by overload of working memory during teaching. Alex Johnstone was there in 1984 (http://pubs.acs.org/doi/abs/10.1021/ed061p847) .
Amongst other issues, he remarks on the amount of “noise” which distracts the student from learning. Sometimes, it is coping with a series of complex instructions while trying to deal with a pipette filler.
Alex died before Christmass at the age of 89. I never met him but I feel I knew him ever since I read about him in the 70 and 80s. This occurred because I tried to gain more CPD in my subject.
Of course people like to extend the triangle to a tetrahedron. Peter Mahaffy call the fourth point the “human element” (http://pubs.acs.org/doi/pdfplus/10.1021/ed083p49)
We are using the word “elements” in a non-chemical context in an essay about education in chemistry. I think Alex would not approve of the word (as it would cause "noise") but approve of the extention because he wrote at the beginning of Chapter 13 of “Energy, Chaos, and Chemical Change” which is entitled “Thermodynamics and Every Things”, “The work in this book will remain largely an academic exercise unless we can see it operating in the world at large.”
Perhaps the word to use is "Relevance".
Here is Alex on syllabus order.
Alex died before Christmass at the age of 89. I never met him but I feel I knew him ever since I read about him in the 70 and 80s. This occurred because I tried to gain more CPD in my subject.
Of course people like to extend the triangle to a tetrahedron. Peter Mahaffy call the fourth point the “human element” (http://pubs.acs.org/doi/pdfplus/10.1021/ed083p49)
We are using the word “elements” in a non-chemical context in an essay about education in chemistry. I think Alex would not approve of the word (as it would cause "noise") but approve of the extention because he wrote at the beginning of Chapter 13 of “Energy, Chaos, and Chemical Change” which is entitled “Thermodynamics and Every Things”, “The work in this book will remain largely an academic exercise unless we can see it operating in the world at large.”
Perhaps the word to use is "Relevance".
Here is Alex on syllabus order.
Of course, any attempt at doing this in England was defeated by the introduction of a KS3 syllabus and exams for 14 year olds (gone, thank goodness!). There still appears a mentality, amongst teachers, that KS3 is still rigidly there even though the exam is now only for 16 year olds. Does anyone teach rganic chemistry to 11 and 12 year olds?
| In this article (http://pubs.acs.org/doi/abs/10.1021/ed077p1571 ) you will find Alex’s 10 commandments of teaching. The 10th is “There should be opportunity given to teach (you don’t really learn until you teach)”. Those not in teaching (especially writers in and to the tabloid press) have the mistaken idea that teachers enter school with all the correct skills to teach and demonstrate practical skills immediately. Teachers are constantly in need of subject and pedagogical CPD and this is at the present being denied. Do notice how wonderfully straightforward his language is. He does not try to wrap up sentences in words which non-researchers would not understand immediately to make the articles appear learned. Remember, to a chemist, “meta” means the third position on the benzene ring; “elements” are our basic building blocks; scaffold is a collection of steel bars with clamps. Just as the student has difficulty in understanding words which have dual (or more) meanings in chemistry and everyday life, so the chemistry teacher has issues with words in psychology which have other meanings in chemistry. |
In a tweet David Read summed Alex's contribution beautifully as “How much of our teaching, and how many of our conversations have been influenced by this giant of Chem Ed? Future generations of Chem Ed practitioners will be influenced by him for decades to come.”
Appendix
Students can quite easily do this and with the CLEAPSS conductivity indicator there is a bonus for for progression in later years.
Can you begin to put some numbers into this experiment
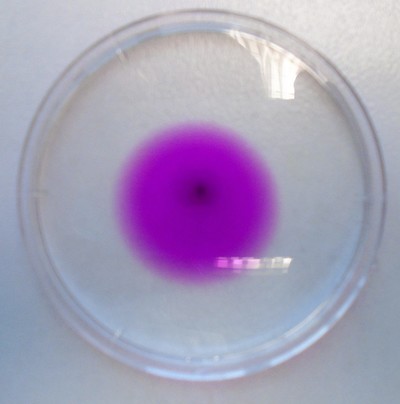
Stir the solution so it is a uniform colour. I would estimate that that crystal has a mass or less than 0.001g. Yet you can see the colour in 10mls of water.
Now to show how you can progress the procedure as students become older and you spiral the curriculum. The second picture shows how a direct electric current distorts the spreading out with the colour moving to the positive electrode so that the purple colour must have a negative charge. This can even be carried out on filter paper dampened with tap (or salt) water. The final picture shows paper with a drop of a mixture of 1 drop of 0.1M potassium chromate, 1 drop of 0.1M copper sulfate in 5 drops of 2M ammonia. The negative chromate ions migrate to the positive electrode and positive copper ions move to the negative electrode. Alex thought the concept of ions, formulae was so difficult in the early stages of learning chemistry that one should always start with organic chemistry, which dealt mainly with atoms and molecules and lots of molecular shapes.
Students can quite easily do this and with the CLEAPSS conductivity indicator there is a bonus for for progression in later years.
- Put 10 ml of cold tap water into a plastic Petri dish.
- Wet the end of a wooden tooth pick in the water, dip the piece tooth pick into a small quantity of potassium manganate(VII) so that a little adheres to the end.
- Place the end in the centre of the dish so the solid comes off the stick.
- What happens with hot water from a kettle
Can you begin to put some numbers into this experiment
Stir the solution so it is a uniform colour. I would estimate that that crystal has a mass or less than 0.001g. Yet you can see the colour in 10mls of water.
- Into a second Petri dish, add 9 ml of water from a measuring cylinder and 1 ml of coloured solution from the first Petri dish, stir gently and decide if the colour can still be seen.
Now to show how you can progress the procedure as students become older and you spiral the curriculum. The second picture shows how a direct electric current distorts the spreading out with the colour moving to the positive electrode so that the purple colour must have a negative charge. This can even be carried out on filter paper dampened with tap (or salt) water. The final picture shows paper with a drop of a mixture of 1 drop of 0.1M potassium chromate, 1 drop of 0.1M copper sulfate in 5 drops of 2M ammonia. The negative chromate ions migrate to the positive electrode and positive copper ions move to the negative electrode. Alex thought the concept of ions, formulae was so difficult in the early stages of learning chemistry that one should always start with organic chemistry, which dealt mainly with atoms and molecules and lots of molecular shapes.